My Alzheimer's Journey
Part 106 - NeuroEPO



Happy Monday!
About a month ago, I heard about a novel Alzheimer’s drug developed in Cuba called NeuroEPO. My digging lead to the College of Medicine at the University of Saskatchewan in Canada. The College of Medicine has partnered with the Center of Molecular Immunology in Cuba for the Phase II trial.
This nasal administered drug began development at the Center of Molecular Immunology in about 2010. I have been unable to learn the exact date. In 2017, a report of the Phase I study was published. This randomized, parallel, open-label study using healthy volunteers was the first human safety evaluation of NeuroEPO.
What is NeuroEPO?
NeuroEPO is a genetically engineered (lab-made) version of erythropoietin (EPO). EPO is a hormone made by your kidneys that tells your bone marrow to make red blood cells. Red blood cells are important because they carry oxygen from your lungs to your brain. Standard EPO is used to treat blood issues like anemia. The research scientists in Cuba believed that an intranasal administration of EPO could benefit patients with neurological disorders - especially, Alzheimer’s disease (AD). The thought is that NeuroEPO could slow the progression of AD.
The 2017 report on the Phase I study stated that NeuroEPO is safe for humans. This was a very small trial though. There were 25 people with 56% being women. Twenty types of adverse effects were noted. Headaches and an increase of hepatic enzymes each made up 20% of the adverse effects. The report stated that “most of the events were mild (95.5%), did not require treatment (88.6%) and were completely resolved (81.8%).”
A later study with a modified formulation called NeuroEPO plus, had very encouraging results. This study was also conducted by the Center for Molecular Immunology, A big difference in this double-blind, randomized, placebo-controlled, Phase II-III trial is that the participants had mild to moderate AD symptoms. The participants of the earlier Phase I trial were in their 20s with no AD symptoms.
The results of the Phase II-III showed:
More than 80% of the clinical trial participants showed their cognitive decline was stopped.
Cognition improved in 54% of the participants.
Over 80% of the control group (received placebo) showed worsening in their cognition.
This is an extraordinary result (if it is true). As for as I know, no drug has ever halted or reversed cognitive decline in humans.
With these amazing results, Cuba partnered with the College of Medicine at the University of Saskatchewan for a clinical trial to test if NeuroEPO improves or maintains cognition in adults with mild to moderate AD. The drug will be administered using a cannula attached to a syringe. The participant will positioned in a Kaiteki position. (the photos below are more or less accurate representations)

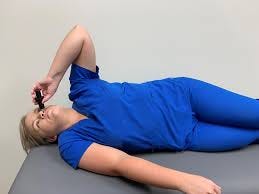
There will be 90 participants (30 placebo and 60 NeuroEPO). NeuroEPO or the placebo will be administered three times a week for 52 weeks.
Interestingly, I was contacted by one of the College of Medicine trial investigators today. He invited me to apply to be a trial participant. The trial is planned to start in a few months. Of course, I would have to go to Saskatchewan. I suspect my Duke Health doctors would have an issue with me participating in this trial.
While researching NeuroEPO, I learned that Cuba has built a very good healthcare system since the United States imposed an embargo on Cuba in 1961. In fact, they develop a lot of their own drugs.
For as long as I can remember, I have wanted to visit Cuba. It has a fascinating culture centered around music and food. Linda and I were beginning to make preliminary plans to visit Cuba. Of course, those plans are on hold due to my AD diagnosis.
The other issue is Trump drooling over Cuba. Maybe those plans will take the same route as Greenland.
I will stay on top of NeuroEPO as the clinical trial gets underway.



Can I ask how he got the medication and if he had any trouble bringing it into the US? I am trying to arrange to take my father to Cuba to get it and want to avoid any issues. I would love to chat if you are able.
Do you have any information on enrollment for the trial in Canada? My brother has EOAD but he lives in the US. Not sure if he would be eligible.